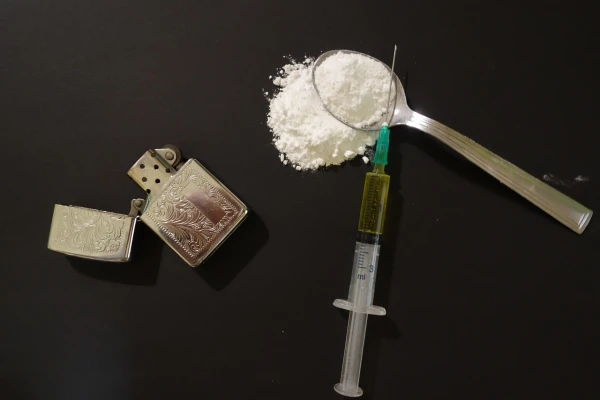
Today, amendments to the law "On the Entry into Force and Application of the Criminal Law" come into effect, which provide for the ban on the synthetic opioid spirchlorphine and products containing it, as well as the establishment of control over a number of other new psychoactive substances, including cyclorphine and the sleeping pill zaleplon, with the definition of their maximum amounts for illegal circulation, reports LETA.
The amendments are designed to prevent the re-emergence of new substances in illegal circulation after the expiration of the temporary ban.
Spirchlorphine is a synthetic opioid analgesic whose effect is two to five times stronger than that of fentanyl, with its maximum allowable amount set at 0.001 grams.
Cyclorphine, in turn, was identified in a study of syringe residues conducted by the Centers for Disease Control and Prevention (CDC) at the end of 2024. Its control is being strengthened by creating a new generic formula that covers other similar compounds to prevent the appearance of such substances on the market.
The bill also includes control over the substance zaleplon, as it can be used for malicious purposes and has not yet been included in the lists of controlled substances. In Latvia, this drug is not registered but has been detected in postal shipment controls.
Given that the substance can be used as a psychoactive, it needs to be included in Appendix 2 of the law – in the third list of narcotic, psychotropic substances and precursors under control in Latvia, which includes dangerous psychotropic substances that can be used for malicious purposes.
The amendments also provide for exceptions from control for a number of substances used in medicine: bupropion, naltrexone, and methylnaltrexone. Bupropion is widely used in the treatment of depression and weight control, it does not cause dependence and has a low potential for abuse. Similarly, naltrexone and methylnaltrexone are prescribed to patients for the treatment of opioid dependence or to reduce side effects caused by them, and the risk of their abuse is extremely low. Exceptions from control prevent additional administrative burdens on doctors and pharmacies, as these drugs will not require special prescription requirements for controlled substances.
According to chemical experts, other substances from the cathinone or amphetamine group can be obtained from bupropion, but at present, such a risk is considered extremely low. The State Agency of Medicines (GAL) actively monitors the sales volumes of these drugs and conducts appropriate monitoring. Thus, if the situation changes, appropriate decisions regarding the control of the substance may be made.
GAL also received a letter from the Latvian Association of Psychiatrists, indicating that there are periodic problems with the availability of medications in the field of psychiatry, including the availability of tablets containing bupropion hydrochloride, which are critically needed for the treatment of certain diseases. To improve the availability of medicines and expedite their acquisition by patients, it is proposed to distribute such drugs as unregistered medicines.
According to statistics, in 2023, about 7,300 packages of medicines containing bupropion were distributed, and in 2024, about 15,300 packages.
Naltrexone hydrochloride is used in combination with other medications or therapy to help overcome opioid dependence. It blocks receptors in the brain, depriving opioids of their effect, so that patients no longer experience euphoria after using them.
Naltrexone does not cause dependence, so the risk of its abuse for intoxication is low. Given the purpose of naltrexone and methylnaltrexone, it is necessary to apply an exception from control to them, as indicated in the annotation to the bill.
Methylnaltrexone-containing medications are used to treat constipation caused by the use of opioid analgesics (e.g., morphine or codeine) of moderate and high strength, in cases where other laxatives are ineffective.
The amendments also clarify and supplement several generic groups. The generic group "dibenzopyrans" has been supplemented to unequivocally include cannabinol and compounds derived from cannabinol or cannabidiol (excluding cannabidiol itself), as well as their isomers and derivatives.
The group "2-phenylmorpholines" has been supplemented with "3-phenylmorpholines" to cover a wider range of potentially dangerous substances.
The law also establishes control "in any quantity" over particularly dangerous substances to eliminate the possibility of their introduction into circulation by mixing with other substances.
In addition, the term "herbal mixture" is replaced with the broader concept of "herbal mass," which also encompasses the upper parts of hemp plants and infused masses. As stated in the annotation to the bill, the term "herbal mixture," according to the expert opinion of the Bureau of Chemical Expertise of the Criminal Police of the State Police, is limiting. The upper parts of hemp plants, processed, for example, with synthetic or semi-synthetic cannabinoids, are often sent for examination.
As previously reported, spirchlorphine is a synthetic opioid, and the use of products containing it can cause serious harm to health, posing a threat to life and public safety as a whole, emphasizes the CDC.
For the manufacture, acquisition, storage, transportation, or shipment for sale, as well as the sale of substances subject to a temporary ban or products containing them, criminal liability is provided – imprisonment for up to two years, forced labor, or a fine and police supervision. If the specified actions lead to serious consequences, the punishment is imprisonment for up to five years or other sanctions provided by law.
The CDC will continue to promptly identify and ban dangerous substances to maximize their harmful use to health and life.












Leave a comment